Xenology: An Introduction to the Scientific Study of Extraterrestrial Life, Intelligence, and Civilization
First Edition
© 1975-1979, 2008 Robert A. Freitas Jr. All Rights Reserved.
Robert A. Freitas Jr., Xenology: An Introduction to the Scientific Study of Extraterrestrial Life, Intelligence, and Civilization, First Edition, Xenology Research Institute, Sacramento, CA, 1979; http://www.xenology.info/Xeno.htm
6.2.3 Towards a Definition of Life
Thermodynamic and statistical principles are among the most fruitful tools of scientific inquiry. They are equally applicable to simple and to complex systems, living or nonliving, terrestrial or extraterrestrial. As Dr. James P. Wesley, Associate Professor of Physics at the University of Missouri in Rolla, tells us: "The relationship of life to the environment is, above all, a thermodynamic relationship. Wherever man may go and whatever alien lifeforms he may encounter, the thermodynamic behavior of life will always be basically predictable."1717 The idea of entropy is often involved in modern discussions of the definition of life.
What is entropy? There are really two relatively straightforward aspects of this concept. The first ties in to the thermodynamic aspects of matter, having to do with heat and energy; the second pertains to statistics and order in any system.
Entropy in the thermodynamic sense is a distinct, physically measurable quantity, much like length, temperature, or weight. At a temperature of absolute zero, to take one example, the entropy in a lump of matter is exactly zero. If the temperature is slowly increased in tiny, reversible little steps, the increase in entropy is mathematically equal to the amount of energy (in joules) divided by the temperature at which it was supplied. This holds even if a change of state occurs, as from solid to liquid.
Suppose that we melt a cube of solid ice at 0 °C. If the mass is 1 kg, the increase in entropy can be calculated as exactly 1223 joules/degree. Entropy in the thermodynamic sense is thus a very real, physical quantity.
In the statistical sense, entropy is a measure of the disorderliness of a system. It seems rather clear that when we melt our 1 kg block of ice, the neat orderly arrangement of water molecules in the cube is destroyed. The rigid crystal lattice is converted into a less ordered system -- the continually changing, sloshing, randomized distribution of molecules in a liquid.
When the orderliness of a system decreases, the entropy correspondingly increases. The situation is analogous to the state of the public library when the shelvers are out on strike. Books are removed from their proper places but are not returned. Disorder and randomness -- entropy -- increase.
The greater the structural complexity of a system, the more information is required to describe it. The more organization a system has, the more information and the less entropy it possesses. But information and orderliness, on the one hand, and entropy, on the other hand, are irreconcilable.
The Second Law of Thermodynamics states that entropy and disorder shall always increase and that information will naturally tend to be degraded and lost in any isolated physical system. (Such isolated systems drift from less probable states to more probable ones.) It is the business of the universe to destroy complexity and to become progressively more randomized.
How does this relate to life? Organisms appear to present a rather curious thermodynamic anomaly. Living systems "violate" the Second Law, by developing well-ordered systems (themselves) out of relatively chaotic systems (their food).85 At first blush, lifeforms seemingly oppose the "universal drive to disorder" mandated by thermodynamic principles. They organize their surroundings and produce order where there was little or none before. Entropy is actually reduced.
This apparent conflict has only been resolved in the last decade or so. Classical treatments dealt with idealized, isolated systems which transfer no energy or matter between themselves and the external environment.2213 In sharp contrast, most systems in nature are nonisolated "open" systems, exchanging matter and energy with the surroundings.
Energy by itself is not enough -- there must be a useful flow of it. This means that to support life, an environment must possess both a "source" and a "sink." Energy emerges from the source, flows to the sink, and is there absorbed.
Living systems customarily establish themselves as intermediate systems, interposed between some source and some sink in the environment. Then, they utilize the energy flow from source to sink to power their own internal functions.
The total entropy of the entire system, which we shall label E, is the sum of the entropies generated in two separate places. First, there is the entropy caused by the source-to-sink energy flow which we shall call S. Then there is the entropy generated by the intermediate system (the living organism) due to exchanges of matter and energy with the surroundings. If we call this L, then we know that the change in total entropy DE = DS + DL.
The second Law of Thermodynamics demands that the total entropy E of any isolated system always increase. Hence, the amount of change must always be positive, so DE > 0. The flow of energy from source to sink (S) consists of irreversible processes, so it too must always cause entropy to increase: DS > 0. Consequently, -DL < DS is our only constraint.
What does this mean in plain English? The last equation above simply says that while the entropy of a living system is always permitted to increase by the Second Law (e.g., upon death), a short range of decrease is allowed as well. That is, it is thermodynamically permissible to have local pockets of negative entropy change -- "negentropy."
Dr. Erwin Schroedinger was really the first to point out that the essence of life is that it feeds on negentropy.1678 An organism able to transfer disorder from itself to its environment can reach a plateau for which the steady-state entropy within the living system is less than the formal entropy entering it.85 Life involves a continuing struggle against increasing entropy.
Living systems thus increase local order at the expense of a larger decrease in order within the environment.
Does life really violate the Second Law of Thermodynamics? We’ve seen that organisms can effect a local decrease in entropy by maintaining an energy flow.* This leads to an ordering of the intermediate (living) system. So the Second Law does not hold for nonisolated systems (L), but only for isolated ones (E). It is invalid for lifeforms alone, but does hold if that same living system is considered in conjunction with the medium in which it is immersed.
Life by itself is a nonisolated system capable of achieving negentropic conditions locally. Life plus environment is an isolated system, for which the total amount of entropy must always increase.
Living systems convert order in their surroundings into disorder, and thereby increase their own internal order. To say that living systems feed on negentropy is equivalent to saying that their existence depends upon increasing the entropy of the rest of the Universe.2213
At the most fundamental level, negentropic ordering processes are achieved by living organisms. Life drives its environment to physical or chemical disequilibrium, establishing an entropy gradient between itself and its surroundings.1144 All living systems possess this feature, and it is contended that any system engaging in such negentropic operations must be considered "living" to a certain extent (Figure 6.4).
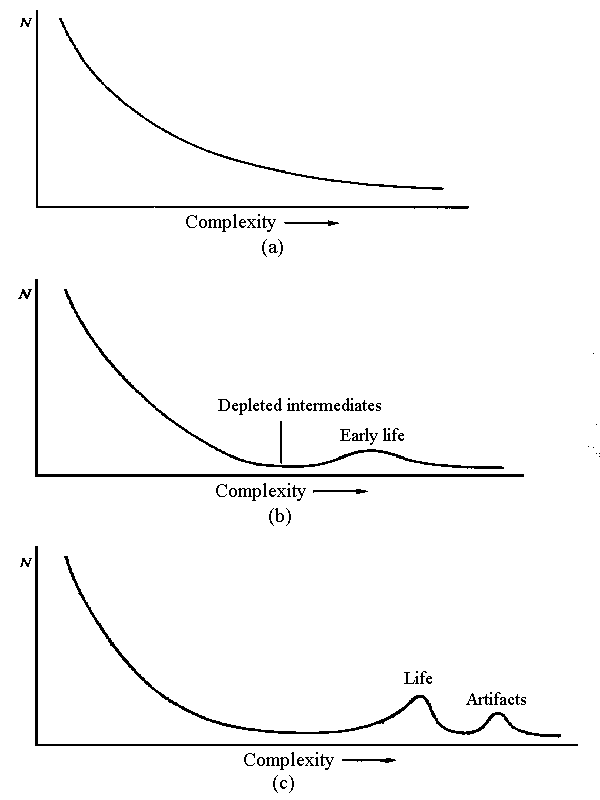
Figure 6.4 Life, Entropy, and Organizational Structure (after Morrison1279)
A schematic presentation of the origins of living things. The first graph shows the population of structures -- molecules, for instance -- against some measure of size or complexity for a case where a free-energy flow has spread the composition to include species of high free energy. The second graph shows what happens when some autocatalytic process begins to increase the population beyond some level of complexity by further degrading the less elaborate, material present. The last graph suggests how artifacts might be looked at in this process, a second and rarer, but more costly, bump.
The question is, of course, to what extent?
Rather than viewing the question of life in absolutist terms, it seems more fruitful to establish the intensity of negentropic processes as a measure of the extent of the life-quality. One of the more fundamental distinctions between "life" and "nonlife" is the degree of organization and internal structure possessed by living systems. Order and structure are virtually synonymous with information content.1012
That is, living systems do more than merely establish a thermodynamic entropy gradient -- they establish an organizational/informational gradient as well. As organisms feed on negentropy, they in effect remove information from the surrounding medium and store it within themselves. It is the business of life to accumulate information and complexity.
In a physical sense, these data bits which permeate all lifeforms may be thought of as being stored in an "aperiodic crystal" -- a biological lattice with highly irregular small-scale nonuniformities.2213 The more effective the negentropic processes, the greater the organization which will arise and hence the more aperiodic the physical structure will become. Organization is maintained by the extraction of order from the environment.
If we consider every "autonegentropic" system to be alive, then its character or richness of expression may be defined along a spectrum from lesser to greater levels of organization. At one extreme are the viruses, which are not negentropic systems by themselves and thus cannot be considered alive in the absence of living hosts. At the other extreme are mules and bees, earlier rejected by the genetic definition of life because of their individual inability to reproduce. These animals are quite clearly auto negentropic systems possessing a vast degree of organization both in the macroscopic and microscopic realms. Thus they are not only "alive" (because they feed on negentropy to build internal complexity) but also "very alive" (because they are so internally complex).**
The refrigerator in my house technically should be considered a "live" system in the very broadest sense, as it is a well-defined intermediate system which uses an energy flow to decrease entropy within (the icebox gets colder, and well-ordered ice crystals collect on the freezer walls) at the expense of increasing the entropy in the external environment (the kitchen air gets warmer). Yet its organizational structure is minimal. Little information is stored, and there is only trivial interrelatedness even on the macroscopic scale. There is scant evidence of aperiodic crystal, no complexity at all on the microscopic stage. So the intensity of life in my refrigerator is negligibly small.
Note that "machine life" or "solid state life" per se is not ruled out. As machines become more and more sophisticated, complexity follows. Large-scale integrated circuits available today pack millions of components onto a tiny silicon wafer the size of a postage stamp. Under the microscope, significant aperiodicities have begun to appear in the latest generation of electronic devices. It is entirely possible that, in time, machines will evolve beyond the point of negligible life-quality. This is true, despite the fact that modern digital computers (which merely process data without adding any of it to their internal structure) are not yet alive at all.
In conclusion, xenologists suspect that there are two fundamental properties any system must possess before it can be considered alive. First, it must be thermodynamically negentropic, establishing an entropy gradient between itself and the environment. Second, it must utilize the entropy gradient to create or to maintain structural order internally -- that is, it must be autonegentropic or self-organizing. Then there is the quality of organization, known as complex interrelatedness or aperiodic crystal, which reflects the intensity of the life process displayed by a given entity.
For those who prefer succinct and pithy definitions, the author would life to offer the following as a starting point for further discussion: Life is negentropic and self-organizing aperiodic crystal.
* A probable corollary is the necessity for "phase separation." In some sense, the sources and sinks should be physically separated with the living system inserted between them. So we expect barriers to exist between an organism’s sources and its sinks. This prevents dissipation of the system, protects it from adverse changes in the environment, and insures the lifeform’s ability to exert and maintain control over its interior.2213 The exact nature of these barriers -- whether gravitational, electromagnetic, or utilizing some hitherto unsuspected principle -- has not been widely discussed.64
** While evolution and the capacity to reproduce are of immense biological importance, a system need not be capable of reproduction for it to be classified as living.2213,62
Last updated on 6 December 2008