Xenology: An Introduction to the Scientific Study of Extraterrestrial Life, Intelligence, and Civilization
First Edition
© 1975-1979, 2008 Robert A. Freitas Jr. All Rights Reserved.
Robert A. Freitas Jr., Xenology: An Introduction to the Scientific Study of Extraterrestrial Life, Intelligence, and Civilization, First Edition, Xenology Research Institute, Sacramento, CA, 1979; http://www.xenology.info/Xeno.htm
7.5 Nucleic Acids and DNA
In the previous section it was mentioned that there are two requirements for the production of proteins. First, there must be amino acids, and second, there must be a way to hook them together to form polymers.
There is, however, a third requirement for the origin of living systems on Earth. It will be recalled from the discussion of the definition of life that "it is the business of life to accumulate information and complexity." Let us consider this mandate in view of the problem of building proteins.
To abiogenetically produce a living system, that system must be capable of accumulating information and order from its environment. The proteins constructed by a cell must have the proper architecture for whatever job needs to be done. So our third requirement may be stated: There must be a way to hook the amino acids together in the correct sequence. Any old proteins will not do -- they must be the right ones.
There exist simple chemical techniques to achieve this kind of ordering. One common example is called "autocatalysis" by chemists. Autocatalysis is a way for a process to catalyze its own production. Once a tiny bit of it has been produced, that bit catalyses the rate of reaction to yield still more, and faster.
Aside from this simple selective feedback effect, the development of molecular self-replication was probably the most critical single event in the origin of life on Earth. The origin of replication and the genetic code, as opposed to the origin of proteins and cells, allowed natural selection to begin to operate on stored information. And once evolution begins, selective advantages of superior membranes and of multicellular colonies can be expressed in the form of increased organismal complexity.
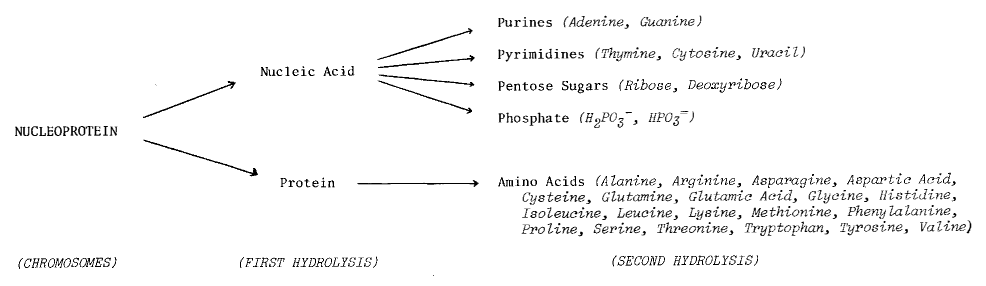
DNA -- the primary information-carrying molecule used by all lifeforms on this planet -- is a polymeric nucleic acid (Figure 7.4). We’ve already seen how easy it is to get amino acids and their polymers. But what about nucleic acids? Can they be demonstrated in prebiotic synthesis experiments, along with their polymers?
Figure 7.4 The Role of Nucleic Acids in Terrestrial Biochemistry
Chemical Structure of Nucleic Acid (from Glasstone72)
DNA Content (figure from Britten and Davidson2568, in Kohne1654)
In 1963, Dr. Cyril Ponnamperuma managed to synthesize adenine (one of the two most important nucleic acid purine bases) under simulated primitive Earth conditions. The NASA scientist and his three colleagues used a Miller-type apparatus, and began their synthesis with nothing more than methane, ammonia and water in the system. The mixture was bombarded with energetic electrons, and about 0.01% of the carbon in the methane was converted into adenine.304 This is highly significant because adenine is useful, not only for making DNA, but also RNA, ATP, ADP, FAD, and a host of other critical life-molecules.
In a related experiment two years later, Dr. John Oró of the University of Houston and A. P. Kimball produced adenine is a closed reaction system which included ammonia, water, and hydrogen cyanide. Heat was supplied as the energy source, and this time the production of the purine base rose to 0.5% of the available carbon.303 This value was observed over a wide range of chemical conditions, indicating the relative ease with which this complex molecule must arise in a plausible prebiotic environment. The synthesis of the other important purine, guanine, has also been convincingly demonstrated.
There have been various attempts to fabricate the three major varieties of pyrimidine bases which are also necessary in the production of nucleic acids. However, the appearance of these substances under conditions similar to the primitive Earth has not been investigated as thoroughly as the purines.
One experiment that yields a hefty 20% of cytosine requires a three-step process involving methane and nitrogen initially to create a cyanoacetylene intermediate, which then goes on to produce the pyrimidine when combined with cyanate ion. Uracil, another pyrimidine, is obtained in very good yield by the direct hydrolysis of cytosine -- a prebiotically reasonable reaction. All the pyrimidines have been synthesized in environments at least arguably analogous to that of the early Earth.
Prebiotic assembly of purines and pyrimidines into full-fledged nucleotides has proven more difficult, and intensive investigations are now underway to determine and eliminate the problem. The main obstacle to success seems to be the formidable complexity of the nucleotide molecules themselves. While bases and sugars are relatively easy to produce, combining them together is a much harder task.
Nevertheless, demonstrations of nucleotide synthesis under geologically plausible constraints have been made. One such technique involves the use of a mediating mineral called apatite, which contains phosphates and oxalate ion, in an "evaporating pond" scenario.
We are not quite home yet. Just as amino acids needed polymerization to become protein, so must nucleotides by polymerized into DNA. What progress has been made in the prebiotic synthesis of polynucleotides?
The experimental record is admittedly spotty. When adenine nucleotides were heated in the presence of polyphosphate for 18 hours at 55 °C, adenine polynucleotide polymers were obtained ranging from 20-30 nucleotides per chain. However, in the words of the experimenter, "the concentration of the reactants had to be as high as possible when the formation of high polymeric material was desired."1625
That is, unless quite artificial conditions were contrived, the adenine nucleotides could not be forged into very long chains. In another experiment, solutions of adenine nucleotide were irradiated with UV light. Long chains were again obtained, but only when extraordinarily high concentrations of polyphosphate were maintained.1628 Under similarly unrealistic conditions, uracil polynucleotides with chain lengths ranging from 10-50 units have been found.1626,1627
One good experiment has been performed by John Oró and E. Stephen-Sherwood, using a plausible "evaporating lakebed" scenario and temperatures from 60-80 °C. Uracil two-unit chains were formed with a yield of 23%, and three-unit segments with a 12% yield. Cytosine polynucleotide chains were obtained by these experimenters with up to six nucleotides in straight-line linkages. Thymine polynucleotides 2-12 units long were produced when an unreasonable chemical environment was used; with more closely matched prebiotic conditions, five-unit chains were obtained in yields of 1% or less.1429
The polymerization of some nucleotides has proven unexpectedly difficult, partly because of the inevitable formation of unnatural side chains and partly because the reaction just doesn’t seem to want to go. Various solutions to these problems have been suggested. For instance, there are enzymes -- ordinary proteins -- that are capable of catalyzing these polymerization reactions with ease. These enzymes, or enzymes like them, could have arisen by nonbiological means. If this is the case, claims one researcher, "such catalysts may have been responsible for the first polymerization of nucleotides on the primitive Earth."72
So at present, here is where we stand. Purines and pyrimidines are comparatively simple to manufacture abiogenetically. The assembly of nucleotides has also met with some limited success, but to date it has proven difficult to synthesize more than six-unit polymeric chains in a prebiotically plausible way.2370
Can these short strands alone make a stab at primitive replication? Dr. Leslie Orgel at the Salk Institute in San Diego, California, mixed up a solution of nucleic acids that might be considered prebiotically reasonable. He then placed some of the six-nucleotide polymers in his specially-enriched "soup." The short-chain DNA polymers correctly replicated themselves once out of every ten tries.
Last updated on 6 December 2008